What Skin Changes Can I Expect With Astaxanthin

Result of Astaxanthin on the Expression and Activity of Aquaporin-3 in Skin in an In-Vitro Study
1
Department of Biomolecular Pharmacology, Hoshi University, two-4-41 Ebara, Shinagawa-ku, Tokyo 142-8501, Japan
two
Department of Clinical Pharmacokinetics, Hoshi Academy, 2-4-41 Ebara, Shinagawa-ku, Tokyo 142-8501, Japan
3
Fuji Chemical Industries Co., Ltd., i Gohkakizawa, Kamiichi-machi, Nakaniikawa-gun, Toyama 930-0405, Japan
four
Department of Functional Molecular Kinetics, Hoshi Academy, 2-iv-41 Ebara, Shinagawa-ku, Tokyo 142-8501, Nihon
*
Authors to whom correspondence should be addressed.
†
These authors contributed equally to this newspaper.
Received: three August 2020 / Revised: one September 2020 / Accustomed: 9 September 2020 / Published: 11 September 2020
Abstruse
Astaxanthin (3,three′-dihydroxy-β,β-carotene-4,four′-dione) is a red lipophilic pigment with strong antioxidant activity. Oral or topical administration of astaxanthin has been reported to amend skin function, including increasing skin moisture. In this study, we examined the mechanism by which astaxanthin improves peel office by focusing on the water channel aquaporin-three (AQP3), which plays important roles in maintaining pare moisture and office. When astaxanthin was added to PHK16-0b or HaCaT cells, the mRNA expression level of AQP3 increased significantly in a concentration-dependent mode in both cell lines. The AQP3 poly peptide expression level was also confirmed to increase when astaxanthin was added to HaCaT cells. Similarly, when astaxanthin was added to 3D human being epidermis model EpiSkin, AQP3 expression increased. Furthermore, when glycerol and astaxanthin were simultaneously added to EpiSkin, glycerol permeability increased significantly compared with that observed for the addition of glycerol lone. We demonstrated that astaxanthin increases AQP3 expression in the skin and enhances AQP3 activity. This consequence suggests that the increased AQP3 expression in the peel is associated with the increase in skin wet by astaxanthin. Thus, we consider astaxanthin useful for treating dry skin caused by decreased AQP3 due to factors such equally diabetes mellitus and aging.
1. Introduction
Astaxanthin (3,3′-dihydroxy-β,β-carotene-four,4′-dione) is a red lipophilic pigment contained in shrimp, salmon, etc. that has potent antioxidant action [1,two]. Astaxanthin ameliorates metabolic diseases, including insulin resistance [3,4] and obesity [5,6], and its usefulness every bit a functional food has been suggested. In addition, there take been reported diverse effects of astaxanthin on the skin [seven,viii,9,x,11,12,thirteen]: increasing pare wet and elasticity, inhibition of wrinkle development, and improving the epidermal barrier. Although these effects on the skin are likely associated with the antioxidant activeness of astaxanthin [fourteen], many unanswered questions remain regarding the underlying mechanism.
Collagens, hyaluronic acid, and ceramides are moisturizing components in the skin, and many cosmetics targeting these substances are commercially available. Forth with these components, the importance of aquaporins (AQPs) in mediating skin moisture is also attracting attending. Thirteen types of AQPs, numbered from AQP0 through AQP12, are expressed in various tissues in humans [fifteen], and AQPs are known as passive transporters of h2o that are vital for h2o homeostasis [16]. Moreover, a subgroup of AQP h2o channels likewise facilitates transmembrane improvidence of pocket-size, polar solutes not only water: AQP3, AQP7, and AQP9 ship glycerol; AQP7, AQP9, and AQP10 send urea; AQP1 transports carbon dioxide and and then on [17,xviii]. Every bit described higher up, AQP is a key molecule that is important for maintaining homeostasis in the living body, and its aberration is known to cause various diseases. Of these AQPs, in the peel, AQP3 is highly expressed in keratinocyte, and a marked decrease in dermal water content and skin elasticity has been reported in AQP3 knockout mice [19,xx,21]. Recently, inquiry has clarified that the AQP3 level in the peel decreases when the skin dries due to psoriasis [22], vitiligo [23,24], diabetes mellitus [25], crumbling [26], etc. We previously confirmed that the dry pare that occurs with the administration of epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors as anticancer agents is associated with decreased AQP3 levels in the skin [27]. Therefore, AQP3 is considered to play important roles in maintaining skin moisture and skin function. In this study, we attempted to analyze the machinery by which astaxanthin improves skin part by focusing on AQP3 in the skin.
2. Materials and Methods
2.one. Materials
Astaxanthin was provided past Fuji Chemic Industries Co., Ltd. (Toyama, Nippon). TRI reagent was purchased from Sigma-Aldrich Corp. (St. Louis, MO, USA). A high-capacity cDNA synthesis kit was purchased from Practical Biosystems (Foster City, CA, United states). RIPA (radioimmunoprecipitation assay) buffer and a protease inhibitor cocktail were purchased from Nacalai Tesque, Inc. (Kyoto, Japan). A rabbit anti-rat AQP3 antibody was purchased from Alomone Labs (Jerusalem, State of israel). A mouse anti-rabbit glyceraldehyde-3-phosphate dehydrogenase (GAPDH) antibody was purchased from Merck Millipore (Darmstadt, Germany). A ass anti-rabbit IgG-HRP (horseradish peroxidase) antibody, sheep anti-mouse IgG-HRP antibody, and enhanced chemiluminescence (ECL) Prime Western blotting detection reagents were purchased from GE Healthcare (Chicago, IL, USA). A glycerol colorimetric assay kit was purchased from Cayman Chemical (Ann Arbor, MI, Usa).
ii.two. PHK16-0b Cell Civilisation
Homo keratinocytes PHK16-0b cells (Health Science Research Resources Depository financial institution, Osaka, Japan) were cultured in MCDB153 medium (5 µg/mL insulin, 0.5 µg/mL hydrocortisone, ten µg/mL transferrin, 0.i mM phosphorylethanolamine, 0.1 mM ethanolamine, 10 ng/mL epithelial growth factor, and 40 µg/mL bovine pituitary excerpt). PHK16-0b cells were plated and maintained in a subconfluent state. After dimethyl sulfoxide (DMSO; final concentration, 0.3%) or astaxanthin (2.five–10 μM) was added, the cells were incubated for 6 h.
2.3. HaCaT Cell Culture
Human keratinocytes HaCaT cells (Cell Lines Service, Eppelheim, Germany) were cultured in DMEM (dulbecco'south modified hawkeye medium) medium (100 μg/mL streptomycin, 100 U/mL penicillin G potassium, and ten% fetal bovine serum). HaCaT cells were plated and maintained in a subconfluent state. After DMSO or astaxanthin (2.5–x μM) was added, the cells were incubated for 6 h or 24 h.
ii.iv. EpiSkin 3D Homo Epidermis Model
The 3D human epidermis model EpiSkin was provided past EpiSkin SNC (Lyon, French republic). EpiSkin was cultured in medium provided past the manufacturer and treated on the upmost side with DMSO or astaxanthin (10 μM) and incubated for 6 h, 24 h, 48 h, or 72 h.
ii.5. Real-Time RT-PCR
RNA was prepared according to the procedure of Chomczynski and Sacchi [28] and chloroform and isopropanol was used for the extraction. A loftier-capacity cDNA synthesis kit was used to synthesize cDNA from 1 μg of RNA. Target gene expression was analyzed by real-time RT-PCR using the primers listed in Table one. Target cistron mRNA expression levels were estimated using the Delta-Delta Ct method and normalized to those of GAPDH. The expression level of GAPDH was stable nether the weather used in this study.
2.6. Preparation of Fractions from HaCaT Cells for Immunoblotting
HaCaT cells were lysed in RIPA buffer supplemented with a protease inhibitor cocktail. The lysate was sonicated and centrifuged (15,000× g for 15 min at four °C). The supernatant was analyzed by Western blotting.
2.vii. Electrophoresis and Western Blotting
An equal amount of loading buffer (100 mM Tris, 20% glycerol, 0.004% bromophenol blue, 4% sodium dodecyl sulfate, and 10% 2-mercaptoethanol; pH 6.viii) was added to the sample solution, and electrophoresed on a 12.5% (w/v) polyacrylamide gel (5 μg protein/lane). The protein was transferred to a polyvinylidene difluoride membrane and blocked with a skim milk solution for 1 h. The membrane was probed with primary antibiotic (one/500; 1 h) and secondary antibody (1/3000; 1 h). Recognized proteins were detected using ECL Prime number detection reagent, and protein immunocomplexes were visualized using a Lumino Image Analyzer.
ii.8. Glycerol Permeability Analysis
EpiSkin was treated on the upmost side with glycerol (final concentration, 100 µM) or a combination of glycerol and astaxanthin (last concentration, 10 µM). Samples were collected from the basal side up to 72 h later on treatment, and the concentration of glycerol was determined using a glycerol colorimetric assay kit.
2.9. Statistical Analysis
Numerical data are expressed as the means ± standard deviations (SDs). Comparisons betwixt two groups were fabricated using a Educatee'south t-test. For comparisons between multiple groups, ANOVA with Bonferroni correction was used.
three. Results
3.1. Event of Astaxanthin on the mRNA Expression Levels of AQP3 in PHK16-0b and HaCaT Cells
Astaxanthin was added to PHK16-0b and HaCaT human keratinocytes, and the mRNA expression levels of AQP3 were measured after 6 h.
When astaxanthin was added to PHK16-0b cells, the mRNA expression level of AQP3 increased significantly in a concentration-dependent way (Figure 1A). Similarly, in HaCaT cells, the mRNA expression level of AQP3 increased in a concentration-dependent manner afterward the addition of astaxanthin. In item, when astaxanthin was added at a concentration of x μM, the mRNA expression of AQP3 increased significantly to a level nearly threefold higher than that in the corresponding control cells (Figure 1B). Nonetheless, 24 h after the improver of astaxanthin to HaCaT cells, the AQP3 mRNA expression level returned to the command level (data not shown).
The in a higher place findings demonstrate that astaxanthin enhances the transcription of AQP3.
3.2. Effect of Astaxanthin on the Protein Expression Level of AQP3 in HaCaT Cells
Astaxanthin was added to HaCaT cells, and the protein expression level of AQP3 was measured after 24 h.
The poly peptide expression level was measured by Western blotting, and the signal bands of AQP3 were detected at approximately 27 kDa and 30 to xl kDa, which have been reported to correspond to nonglycosylated (27 kDa) and glycosylated (30 to 40 kDa) AQP3 [29,xxx]. The presence or absence of glycosylation results in differences in AQP stability and migration to the cell membrane but does non affect their water permeability functions [31,32,33]. Therefore, in this study, the sum of these bands was analyzed as the expression level of AQP3. When astaxanthin was added to HaCaT cells, the poly peptide expression of AQP3 increased significantly to a level almost twofold higher than that in the respective command cells (Figure 2).
The higher up findings demonstrate that astaxanthin increases the poly peptide expression level, in addition to the mRNA expression level, of AQP3.
3.three. Upshot of Astaxanthin on Glycerol Permeability via AQP3 in the EpiSkin Epidermal System
AQP3 transports glycerol in improver to water [34]. We assessed the effect of astaxanthin on glycerol permeability via AQP3 in the EpiSkin epidermal organization. The EpiSkin epidermal arrangement is a reconstituted organotypic culture of human being keratinocytes forming a multilayer differentiated epidermis on a collagen matrix [35,36].
When astaxanthin was added to EpiSkin, the mRNA expression of AQP3 increased significantly, to a level nigh 2.5-fold higher than that in the control group (Effigy 3A). Similar results were obtained when astaxanthin was added to PHK16-0b and HaCaT cells. Simultaneous addition of glycerol and astaxanthin to EpiSkin significantly increased glycerol permeability compared with that resulting from the add-on of glycerol alone (Figure 3B).
These results demonstrate that astaxanthin increases the expression of AQP3 in EpiSkin and enhances glycerol permeability.
4. Conclusions
Water channel AQPs have been validated as an important drug target but there is no unmarried drug that has yet been approved to successfully target them [37,38]. Therefore, the search for a drug targeting AQP is very important, and much enquiry has been done in the world. Oral or topical administration of astaxanthin has been reported to improve peel function, including an increment in skin moisture [seven,8,9,10,xi,12,13]. In this report, we examined the mechanism by which astaxanthin improves skin function by focusing on AQP3, which plays of import roles in maintaining skin wet and office.
PHK16-0b is an immortalized foreskin epidermal keratinocyte cell line transformed with human papilloma virus (HPV) 16 and which expresses AQP3 [39,forty]. HaCaT cells are a transformed immortal keratinocyte cell line used widely in scientific research on AQP3 [41,42]. When astaxanthin was added to PHK16-0b and HaCaT cells, the mRNA expression level of AQP3 increased significantly in a concentration-dependent fashion in both cell lines (Figure 1). In addition, an increase in the protein expression level of AQP3 was besides confirmed (Figure 2). Furthermore, astaxanthin increased glycerol permeability in the EpiSkin epidermal arrangement (Figure iii). These results suggest that astaxanthin enhances AQP3 activity through an increase in AQP3 expression. As previous studies take reported a decrease in pare moisture in AQP3 knockout mice [xix,20,21], substances that increase AQP3 in the skin are considered to be useful as skin moisturizers. The present study suggests that the increased expression and activity of AQP3 is involved in the skin moisturizing action of astaxanthin, in addition to its antioxidant effect [14].
Astaxanthin acts as a peroxisome proliferator-activated receptor-gamma (PPARγ) modulator and induces the expression of liver X receptor (LXR) via PPARγ activation [43,44]. In improver, recent studies take demonstrated that ligands of either PPARγ or LXR increment the expression level of AQP3 in epidermal keratinocytes [45,46,47]. These results suggest that astaxanthin enhances the transcription of AQP3 via its PPARγ agonist action and increases AQP3 protein expression, thereby enhancing AQP3 action.
The rate of carotenoid assimilation from the digestive tract is depression. Yet, oral assistants of astaxanthin to mice was reported to increment both the blood concentration and skin content of astaxanthin [13]. Thus, we believe that the demonstrated effects of the increased AQP3 expression level in the peel volition occur with either oral or topical administration.
As mentioned above, AQP has subfamilies and is widely distributed throughout the body. It was examined whether the AQP3 increasing effect past astaxanthin was similarly observed on cells of other tissues. Every bit a outcome, it was plant that astaxanthin significantly increased the mRNA expression level of AQP3 in human colon cancer HT-29 cell line (data non shown). AQP3 in the large intestine is important for regulation of fecal water content [48]. Therefore, information technology was considered that astaxanthin may be a useful substance for abnormal h2o metabolism in the digestive tract.
Information technology was reported that the AQP3 level in the skin decreases when the skin dries due to psoriasis [22], vitiligo [23,24], diabetes mellitus [25], crumbling [26], and anti-cancer drugs [27]. This effect suggests that the increased AQP3 expression in the skin is associated with the increase in skin wet by astaxanthin. Thus, we consider astaxanthin useful for treating dry out skin caused by decreased AQP3 due to factors such equally diabetes mellitus and aging etc. Further developments in this research surface area are expected.
Author Contributions
Conceptualization, N.I., J.T. and M.S.; methodology, N.I., R.K., J.T. and G.S.; formal analysis, N.I., R.K., C.N., A.I. and Y.S.; writing—original typhoon preparation, N.I., R.G., and A.I.; writing—review and editing, K.S.; projection assistants, 1000.S. All authors accept read and agreed to the published version of the manuscript.
Funding
This research was supported by the Fuji Chemical Industries Co., Ltd.
Conflicts of Interest
The authors declare no disharmonize of interest. The funders had no role in the design of the study; in the collection, analyses, or estimation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Naguib, Y.Thou.A. Antioxidant Activities of Astaxanthin and Related Carotenoids. J. Agric. Food Chem. 2000, 48, 1150–1154. [Google Scholar] [CrossRef] [PubMed]
- Ohgami, K.; Shiratori, 1000.; Kotake, S.; Nishida, T.; Mizuki, Northward.; Yazawa, Thousand.; Ohno, S. Effects of Astaxanthin on Lipopolysaccharide-Induced Inflammation in vitro and in vivo. Investig. Opthalmol. Vis. Sci. 2003, 44, 2694–2701. [Google Scholar] [CrossRef] [PubMed]
- Hussein, G.; Nakagawa, T.; Goto, H.; Shimada, Y.; Matsumoto, K.; Sankawa, U.; Watanabe, H. Astaxanthin Ameliorates Features of Metabolic Syndrome in SHR/NDmcr-cp. Life Sci. 2007, 80, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Nagashimada, M.; Zhuge, F.; Zhan, L.; Nagata, N.; Tsutsui, A.; Nakanuma, Y.; Kaneko, S.; Ota, T. Astaxanthin Prevents and Reverses Diet-Induced Insulin Resistance and Steatohepatitis in Mice: A Comparison with Vitamin E. Sci. Rep. 2015, 5, 17192. [Google Scholar] [CrossRef]
- Ikeuchi, Grand.; Koyama, T.; Takahashi, J.; Yazawa, Yard. Effects of Astaxanthin in Obese Mice Fed a Loftier-Fat Nutrition. Biosci. Biotechnol. Biochem. 2007, 71, 893–899. [Google Scholar] [CrossRef]
- Wang, J.; Liu, Due south.; Wang, H.; Xiao, S.; Li, C.; Li, Y.; Liu, B. Xanthophyllomyces Dendrorhous-Derived Astaxanthin Regulates Lipid Metabolism and Gut Microbiota in Obese Mice Induced by A Loftier-Fat Diet. Mar. Drugs 2019, 17, 337. [Google Scholar] [CrossRef]
- Chalyk, N.E.; Klochkov, V.A.; Bandaletova, T.Y.; Kyle, N.H.; Petyaev, I.M. Continuous Astaxanthin Intake Reduces Oxidative Stress and Reverses Age-Related Morphological Changes of Rest Skin Surface Components in Middle-Aged Volunteers. Nutr. Res. 2017, 48, xl–48. [Google Scholar] [CrossRef]
- Guerin, M.; Huntley, M.E.; Olaizola, M. Haematococcus Astaxanthin: Applications for Human Health and Nutrition. Trends Biotechnol. 2003, 21, 210–216. [Google Scholar] [CrossRef]
- Hama, Southward.; Takahashi, K.; Inai, Y.; Shiota, K.; Sakamoto, R.; Yamada, A.; Tsuchiya, H.; Kanamura, K.; Yamashita, E.; Kogure, K. Protective Effects of Topical Application of a Poorly Soluble Antioxidant Astaxanthin Liposomal Formulation on Ultraviolet-Induced Skin Damage. J. Pharm. Sci. 2012, 101, 2909–2916. [Google Scholar] [CrossRef]
- Ng, Q.10.; De Deyn, Chiliad.L.Z.Q.; Loke, Westward.; Foo, N.X.; Chan, H.W.; Yeo, W.Southward. Furnishings of Astaxanthin Supplementation on Peel Health: A Systematic Review of Clinical Studies. J. Nutrition. Suppl. 2020, 1–14. [Google Scholar] [CrossRef]
- Tominaga, K.; Hongo, N.; Karato, M.; Yamashita, E. Cosmetic Benefits of Astaxanthin on Humans Subjects. Acta Biochim. Pol. 2012, 59, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Edwin, D.L. Equol'southward Effecacy is Greater than Astaxanthin for Antioxidants, Extracellular Matrix Integrity & Breakup, Growth Factors and Inflammatory Biomarkers via Human Skin Gene Expression Analysis. J. Funct. Foods 2019, 59, 380–393. [Google Scholar]
- Komatsu, T.; Sasaki, S.; Manabe, Y.; Hirata, T.; Sugawara, T. Preventive Event of Dietary Astaxanthin on UVA-Induced Pare Photoaging in Hairless Mice. PLoS One 2017, 12, e0171178. [Google Scholar] [CrossRef] [PubMed]
- Davinelli, S.; Nielsen, M.Due east.; Scapagnini, Thousand. Astaxanthin in Skin Health, Repair, and Illness: A Comprehensive Review. Nutrients 2018, 10, 522. [Google Scholar] [CrossRef]
- Fujiyoshia, Y.; Mitsuoka, K.; De Groot, B.L.; Philippsen, A.; Grubmüller, H.; Agre, P.; Engel, A. Structure and Function of H2o Channels. Curr. Opin. Struct. Biol. 2002, 12, 509–515. [Google Scholar] [CrossRef]
- Kitchen, P.; 24-hour interval, R.E.; Salman, M.Chiliad.; Conner, Grand.T.; Neb, R.G.; Conner, A.C. Beyond Water Homeostasis: Various Functional Roles of Mammalian Aquaporins. Biochim. Biophys. Acta (BBA) Gen. Subj. 2015, 1850, 2410–2421. [Google Scholar] [CrossRef]
- Kitchen, P.; Salman, M.Chiliad.; Pickel, S.U.; Jennings, J.; Törnroth-Horsefield, S.; Conner, Grand.T.; Bill, R.M.; Conner, A.C. Water Aqueduct Pore Size Determines Exclusion Properties merely not Solute Selectivity. Sci. Rep. 2019, 9, i–14. [Google Scholar] [CrossRef]
- Hara-Chikuma, M.; Verkman, A.S. Physiological Roles of Glycerol-Transporting Aquaporins: The Aquaglyceroporins. Cell. Mol. Life Sci. 2006, 63, 1386–1392. [Google Scholar] [CrossRef]
- Hara, M.; Ma, T.; Verkman, A.S. Selectively Reduced Glycerol in Skin of Aquaporin-3-Deficient Mice May Account for Dumb Skin Hydration, Elasticity, and Barrier Recovery. J. Biol. Chem. 2002, 277, 46616–46621. [Google Scholar] [CrossRef]
- Ma, T.; Hara, M.; Sougrat, R.; Verbavatz, J.-M.; Verkman, A.S. Impaired Stratum Corneum Hydration in Mice Lacking Epidermal Water Aqueduct Aquaporin-three. J. Biol. Chem. 2002, 277, 17147–17153. [Google Scholar] [CrossRef]
- Qin, H.; Zheng, X.; Zhong, X.; Shetty, A.K.; Elias, P.K.; Bollag, West.B. Aquaporin-iii in Keratinocytes and Peel: Its Role and Interaction with Phospholipase D2. Arch. Biochem. Biophys. 2011, 508, 138–143. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Je, Y.-J.; Lee, Southward.-Southward.; Li, Z.J.; Choi, D.-One thousand.; Kwon, Y.-B.; Sohn, Chiliad.-C.; Im, 1000.; Seo, Y.J.; Lee, J.H. Changes in Transepidermal Water Loss and Skin Hydration According to Expression of Aquaporin-3 in Psoriasis. Ann. Dermatol. 2012, 24, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.-H.; Lee, A.-Y. Reduced Aquaporin3 Expression and Survival of Keratinocytes in the Depigmented Epidermis of Vitiligo. J. Investig. Dermatol. 2010, 130, 2231–2239. [Google Scholar] [CrossRef]
- Lee, A.-Y. Office of Keratinocytes in the Development of Vitiligo. Ann. Dermatol. 2012, 24, 115–125. [Google Scholar] [CrossRef]
- Ikarashi, Northward.; Mizukami, N.; Kon, R.; Kaneko, M.; Uchino, R.; Fujisawa, I.; Fukuda, N.; Sakai, H.; Kamei, J. Study of the Mechanism Underlying the Onset of Diabetic Xeroderma Focusing on an Aquaporin-3 in a Streptozotocin-Induced Diabetic Mouse Model. Int. J. Mol. Sci. 2019, 20, 3782. [Google Scholar] [CrossRef]
- Ikarashi, N.; Kon, R.; Kaneko, Yard.; Mizukami, North.; Kusunoki, Y.; Sugiyama, K. Human relationship between Aging-Related Peel Dryness and Aquaporins. Int. J. Mol. Sci. 2017, 18, 1559. [Google Scholar] [CrossRef]
- Ikarashi, Due north.; Kaneko, M.; Watanabe, T.; Kon, R.; Yoshino, Thousand.; Yokoyama, T.; Tanaka, R.; Takayama, Northward.; Sakai, H.; Kamei, J. Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor Erlotinib Induces Dry Skin via Decreased in Aquaporin-iii Expression. Biomolecules 2020, 10, 545. [Google Scholar] [CrossRef] [PubMed]
- Chomczynski, P.; Sacchi, Due north. Single-step Method of RNA Isolation by Acid Guanidinium Thiocyanate-Phenol-Chloroform Extraction. Anal. Biochem. 1987, 162, 156–159. [Google Scholar] [CrossRef]
- Silberstein, C.; Kierbel, A.; Amodeo, G.; Zotta, East.; Bigi, F.; Berkowski, D.; Ibarra, C. Functional Characterization and Localization of AQP3 in the Man Colon. Braz. J. Med. Biol. Res. 1999, 32, 1303–1313. [Google Scholar] [CrossRef]
- Spector, D.A.; Wade, J.B.; Dillow, R.; Steplock, D.A.; Weinman, E.J. Expression, Localization, and Regulation of Aquaporin-1 to -3 in Rat Urothelia. Am. J. Physiol. Physiol. 2002, 282, F1034–F1042. [Google Scholar] [CrossRef]
- Baumgarten, R.; Van De Pol, Grand.H.; Wetzels, J.F.; Van Bone, C.H.; Deen, P.M. Glycosylation is not Essential for Vasopressin-Dependent Routing of Aquaporin-2 in Transfected Madin-Darby Canine Kidney Cells. J. Am. Soc. Nephrol. 1998, 9, 1553–1559. [Google Scholar] [PubMed]
- Hendriks, G.; Wang, F.; Thirumurugan, K.; Stafford, W.F.; Hammer, J.A.; Knight, P.J.; Sellers, J.R.; Koudijs, M.; Van Balkom, B.Westward.M.; Oorschot, 5.; et al. Glycosylation Is Important for Cell Surface Expression of the Water Channel Aquaporin-ii but Is Non Essential for Tetramerization in the Endoplasmic Reticulum. J. Biol. Chem. 2003, 279, 2975–2983. [Google Scholar] [CrossRef] [PubMed]
- Umenishi, F.; Narikiyo, T.; Schrier, R.Westward. Event on Stability, Degradation, Expression, and Targeting of Aquaporin-two H2o Channel past Hyperosmolality in Renal Epithelial Cells. Biochem. Biophys. Res. Commun. 2005, 338, 1593–1599. [Google Scholar] [CrossRef]
- Ikarashi, N.; Nagoya, C.; Kon, R.; Kitaoka, S.; Kajiwara, Due south.; Saito, M.; Kawabata, A.; Ochiai, W.; Sugiyama, K. Changes in the Expression of Aquaporin-3 in the Gastrointestinal tract Affect Drug Absorption. Int. J. Mol. Sci. 2019, 20, 1559. [Google Scholar] [CrossRef] [PubMed]
- Luu-The, V.; Duche, D.; Ferraris, C.; Meunier, J.-R.; LeClaire, J.; Labrie, F. Expression Profiles of Phases 1 and 2 Metabolizing Enzymes in Human Skin and the Reconstructed Skin Models Episkin™ and Full Thickness Model from Episkin™. J. Steroid Biochem. Mol. Biol. 2009, 116, 178–186. [Google Scholar] [CrossRef]
- Roguet, R.; Cohen, C.; Dossou, K.; Rougier, A. Episkin, a Reconstituted Human Epidermis for Assessing in vitro the Irritancy of Topically Practical Compounds. Toxicol. Vitro 1994, viii, 283–291. [Google Scholar] [CrossRef]
- Abir-Awan, M.; Kitchen, P.; Salman, M.M.; Conner, M.; Conner, A.C.; Bill, R.M. Inhibitors of Mammalian Aquaporin Water Channels. Int. J. Mol. Sci. 2019, twenty, 1589. [Google Scholar] [CrossRef]
- Verkman, A.S.; Anderson, M.O.; Papadopoulos, M.C. Aquaporins: Important but Elusive Drug Targets. Nat. Rev. Drug Discov. 2014, 13, 259–277. [Google Scholar] [CrossRef]
- Hashida, T.; Yasumoto, Due south. Induction of Chromosome Abnormalities in Mouse and Man Epidermal Keratinocytes past the Human Papillomavirus Type 16 E7 Oncogene. J. Gen. Virol. 1991, 72, 1569–1577. [Google Scholar] [CrossRef]
- Ikarashi, N.; Ogiue, N.; Toyoda, E.; Nakamura, Chiliad.; Kon, R.; Kusunoki, Y.; Aburada, T.; Ishii, Thou.; Tanaka, Y.; Machida, Y.; et al. Elucidating the Mechanism past Which Gypsum fibrosum, a Traditional Chinese Medicine, Maintains Cutaneous Water Content. Biol. Pharm. Bull. 2013, 36, 1615–1621. [Google Scholar] [CrossRef]
- Varma, S.R.; Sivaprakasam, T.O.; Arumugam, I.; Dilip, N.; Raghuraman, M.; Pavan, K.; Rafiq, 1000.; Paramesh, R. In vitro Anti-Inflammatory and Skin Protective Backdrop of Virgin Coconut Oil. J. Tradit. Complement. Med. 2019, nine, 5–xiv. [Google Scholar] [CrossRef]
- Xing, F.; Liao, West.; Jiang, P.; Xu, W.; Jin, 10. Issue of Retinoic Acid on Aquaporin 3 Expression in Keratinocytes. Genet. Mol. Res. 2016, fifteen, 15016951. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.-I. Astaxanthin as a Peroxisome Proliferator-Activated Receptor (PPAR) Modulator: Its Therapeutic Implications. Mar. Drugs 2019, 17, 242. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Tanabe, H.; Matsumoto, A.; Takagi, M.; Umegaki, K.; Amagaya, Due south.; Takahashi, J. Astaxanthin Functions Differently as a Selective Peroxisome Proliferator-Activated Receptor Gamma Modulator in Adipocytes and Macrophages. Biochem. Pharmacol. 2012, 84, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.J.; Kim, P.; Lu, Y.F.; Feingold, K.R. PPARgamma Activators Stimulate Aquaporin 3 Expression in Keratinocytes/Epidermis. Exp. Dermatol. 2011, 20, 595–599. [Google Scholar] [CrossRef]
- Schmuth, Thou.; Moosbrugger-Martinz, V.; Blunder, South.; Dubrac, S. Role of PPAR, LXR, and PXR in Epidermal Homeostasis and Inflammation. Biochim. Biophys. Acta (BBA) Mol. Cell Biol. Lipids 2014, 1841, 463–473. [Google Scholar] [CrossRef]
- Yang, R.; Chowdhury, Due south.; Choudhary, V.; Chen, 10.; Bollag, W. Keratinocyte Aquaporin-3 Expression Induced by Histone Deacetylase Inhibitors is Mediated in Role by Peroxisome Proliferator-Activated Receptors (PPARs). Exp. Dermatol. 2020, 29, 380–386. [Google Scholar] [CrossRef]
- Ikarashi, N.; Kon, R.; Sugiyama, Thousand. Aquaporins in the Colon every bit a New Therapeutic Target in Diarrhea and Constipation. Int. J. Mol. Sci. 2016, 17, 1172. [Google Scholar] [CrossRef]
Figure 1. Result of astaxanthin on the mRNA expression level of aquaporin-iii (AQP3) in PHK16-0b (A) and HaCaT (B) cells. Astaxanthin was added to PHK16-0b or HaCaT cells, and the cells were incubated for 6 h. The AQP3 mRNA expression level was measured past real-fourth dimension RT-PCR. Expression levels were normalized to those of glyceraldehyde-3-phosphate dehydrogenase (GAPDH), and the information are presented equally the hateful values as a percent of the command values, which were prepare to 100% (mean ± SD, due north = 4; ** p < 0.01 and *** p < 0.001).
Figure one. Effect of astaxanthin on the mRNA expression level of aquaporin-3 (AQP3) in PHK16-0b (A) and HaCaT (B) cells. Astaxanthin was added to PHK16-0b or HaCaT cells, and the cells were incubated for 6 h. The AQP3 mRNA expression level was measured by existent-time RT-PCR. Expression levels were normalized to those of glyceraldehyde-iii-phosphate dehydrogenase (GAPDH), and the data are presented as the mean values as a percentage of the control values, which were set to 100% (mean ± SD, n = 4; ** p < 0.01 and *** p < 0.001).
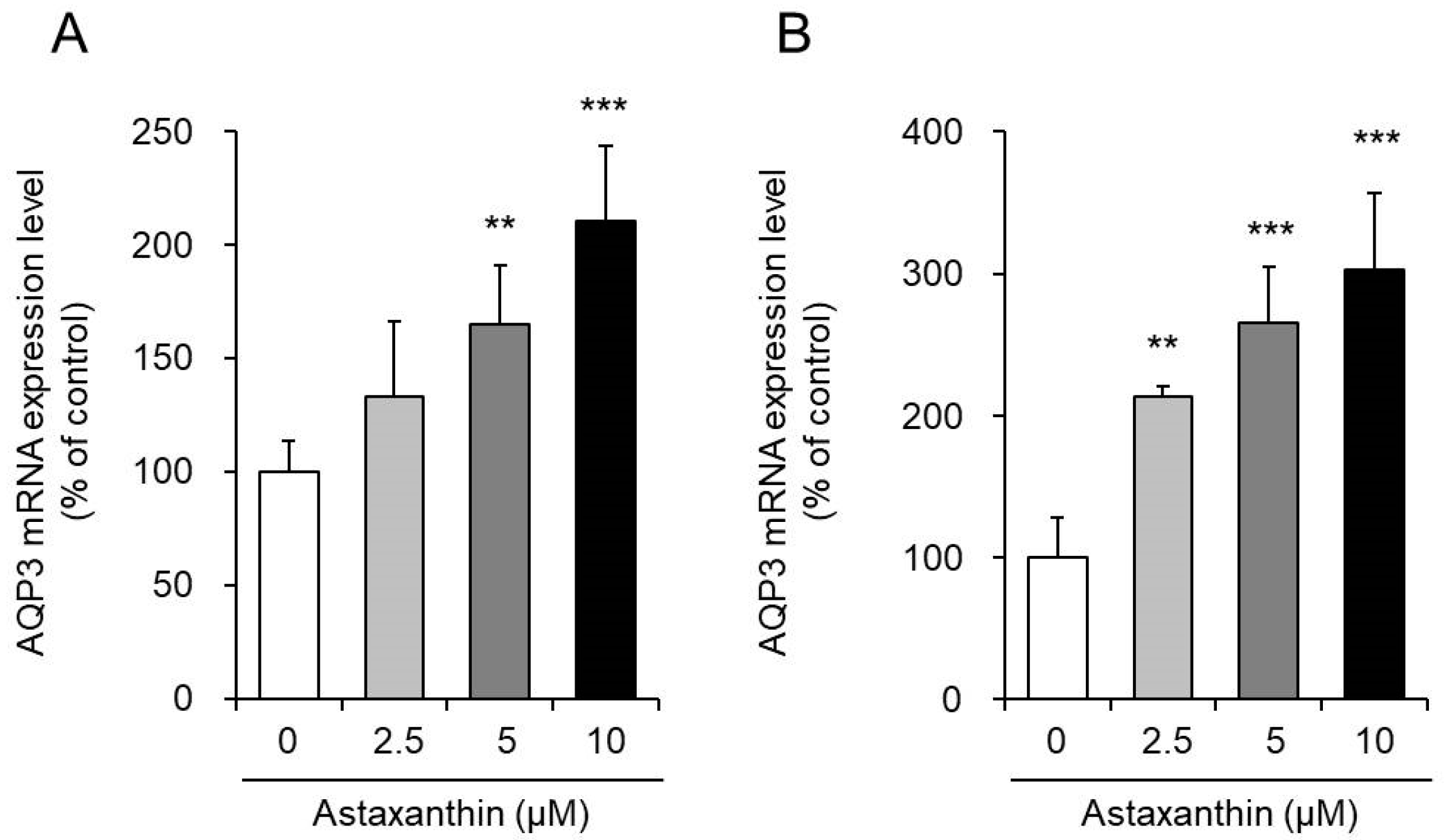
Figure 2. Effect of astaxanthin on the protein expression level of AQP3 in HaCaT cells. Astaxanthin was added to HaCaT cells, and the cells were incubated for 24 h. The AQP3 protein expression in HaCaT cells was analyzed by Western blotting. Expression levels were normalized to those of GAPDH, the data are shown equally the mean values as a percentage of the control values, which were set to 100% (mean ± SD, n = five; ** p < 0.01).
Effigy 2. Effect of astaxanthin on the protein expression level of AQP3 in HaCaT cells. Astaxanthin was added to HaCaT cells, and the cells were incubated for 24 h. The AQP3 protein expression in HaCaT cells was analyzed by Western blotting. Expression levels were normalized to those of GAPDH, the data are shown as the hateful values as a percentage of the control values, which were set to 100% (mean ± SD, n = 5; ** p < 0.01).
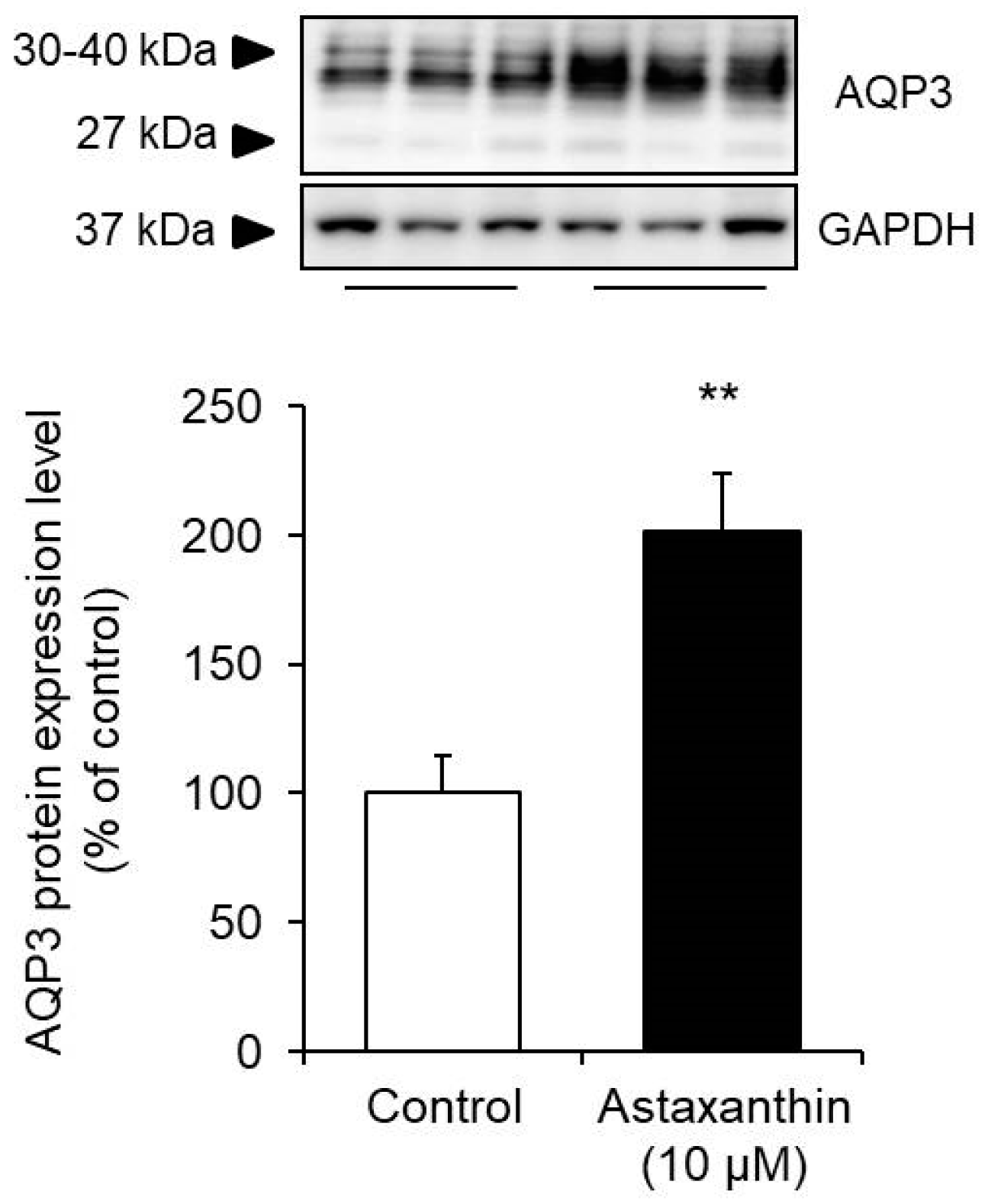
Figure iii. Effect of astaxanthin on glycerol permeability via AQP3. (A) Astaxanthin was added to EpiSkin and incubated for 6 h. The AQP3 mRNA expression level was measured by existent-time RT-PCR. The expression level was normalized to that of GAPDH, and the data are presented as the hateful value equally a percentage of the command value, which was set to 100%. (B) EpiSkin was treated on the apical side with glycerol or a combination of glycerol and astaxanthin. Samples were collected from the basal side 24 h, 48 h, and 72 h subsequently treatment, and the concentration of glycerol was adamant (hateful ± SD, n = 6; * p < 0.05 and ** p < 0.01).
Figure 3. Effect of astaxanthin on glycerol permeability via AQP3. (A) Astaxanthin was added to EpiSkin and incubated for 6 h. The AQP3 mRNA expression level was measured by real-fourth dimension RT-PCR. The expression level was normalized to that of GAPDH, and the data are presented as the mean value as a percentage of the control value, which was set up to 100%. (B) EpiSkin was treated on the apical side with glycerol or a combination of glycerol and astaxanthin. Samples were nerveless from the basal side 24 h, 48 h, and 72 h later on treatment, and the concentration of glycerol was determined (mean ± SD, n = six; * p < 0.05 and ** p < 0.01).
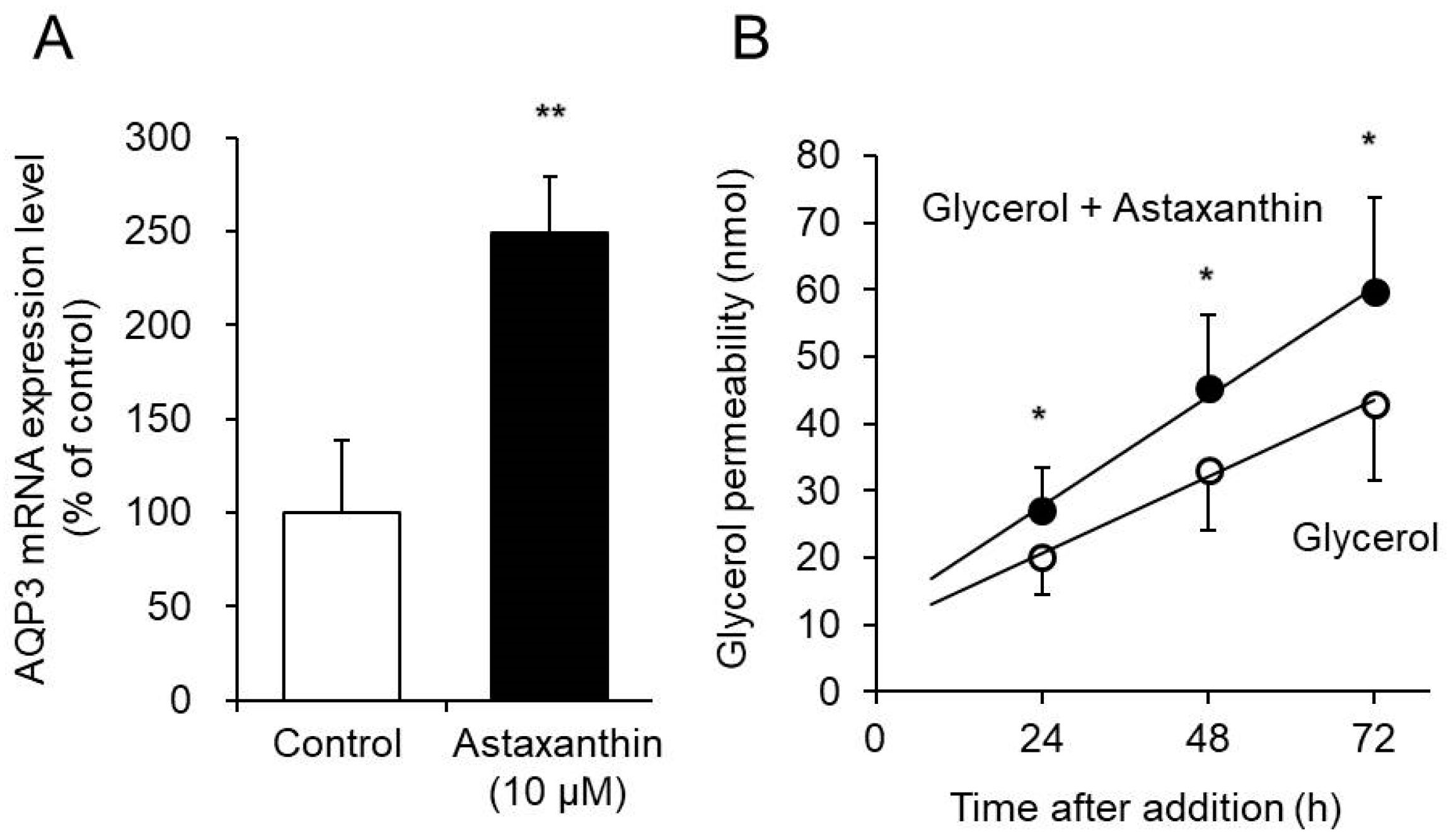
Table 1. Primer sequences used for existent-fourth dimension PCR.
Table 1. Primer sequences used for real-time PCR.
| Target | Frontward Primer (5′ to three′) | Reverse Primer (v′ to 3′) |
|---|---|---|
| AQP3 | AGACAGCCCCTTCAGGATTT | TCCCTTGCCCTGAATATCTG |
| GAPDH | ATGGGGAAGGTGAAGGTCG | GGGGTCATTGATGGCAACAATA |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open admission article distributed under the terms and conditions of the Creative Eatables Attribution (CC BY) license (http://creativecommons.org/licenses/by/four.0/).
Source: https://www.mdpi.com/2075-1729/10/9/193/htm
Posted by: ruffnertoorepithe1949.blogspot.com

0 Response to "What Skin Changes Can I Expect With Astaxanthin"
Post a Comment